Novel Coronavirus disease 2019, commonly referred to as COVID-19, has affected almost every country in the world, and mankind is scrambling to find sustainable preventive and curative solutions for this condition.
There is no specific antiviral therapy available to treat COVID-19 patients and this is leading to loss of a small proportion of lives on a daily basis. There have been numerous attempts at research and numerous trials are underway in finding treatment modalities for the ongoing pandemic COVID-19.

One interesting modality that has attracted global attention and is under evaluation for its effectiveness could be the “convalescent plasma therapy”, which involves transferring the antibodies from a person who has recovered from the coronavirus disease to a critically-ill patient, to help boost their immunity.
Convalescent plasma therapy has been found to be safe and effective against SARS, MERS, and H1N1 viral infections over the past two decades and the same hypotheses are being applied for SARS-COV-2 infection that belongs to the same class of viruses (corona).
What is Convalescent Plasma?
Human blood comprises two main components – the cells and the fluid. Plasma refers to the fluid component of blood. Proteins like antibodies are present in the plasma. People who have recovered from COVID-19 have antibodies against it in their plasma and this refers to ‘convalescent plasma’.
Researchers continue to study the use of plasma of COVID-19 recovered patients to treat existing COVID-19 patients. Plasma extracted from blood donated by recovered patients is used for convalescent plasma therapy.
How Does Convalescent Plasma Therapy Work?
Whenever an individual gets infected by a new pathogen like a virus or bacteria for the first time, his/her body’s immune system gets activated and it tries to kill the invading agents by various means.
One of the mechanisms involves the production of proteins called ‘antibodies’ that are released by certain immune system cells in the blood. When these cells come in contact with the new pathogen then they produce certain specific proteins called antibodies that carry identity marks specific to that particular pathogen and help the immune system recognise and locate those pathogens in the body.
These antibodies travel around the body in the bloodstream and whenever a pathogen comes in contact with a matching antibody, it raises a sort of an alarm leading to a cascade of immune response wherein the body’s immune system tries to kill the invading pathogen.
Convalescent plasma of people who recovered from COVID-19 is found to contain antibodies against the novel coronavirus and researchers are trying to see if transfusing convalescent plasma helps existing patients recover from the disease.
The antibodies in the plasma from a recovered COVID-19 patient is expected to enable the immune system to identify and fight the coronavirus infection.
Is convalescent plasma effective for the treatment of COVID‐19 infection in cancer patients?
There is no strong data from scientific studies supporting the use of convalescent plasma in the treatment of COVID19. Data from some studies suggest that convalescent plasma with high antibody levels may lessen the severity or shorten the duration of COVID-19 in some people when given early in the disease or in those with weakened immune systems.
Studies have shown no benefit in terms of reducing severity of disease or early recovery or reducing risk of death when used in hospitalised patients. When given early in the course of disease (within 3-4days of symptom onset), a few studies show benefit in terms of early recovery. The evidence is not strong enough to recommend for or against its use in various stages of disease.
Convalescent plasma therapy might at least help some patients recover, or may lessen the severity of the illness, or may shorten the length of the disease. However, we still don’t know which group of patients are likely to benefit and whether it may work in all cancer patients or not.
Is convalescent plasma safe for the treatment of COVID‐19 infection in cancer patients?
Blood and blood products have been used to treat many other conditions and it’s usually very safe. Studies have shown no major adverse effects with use of convalescent plasma.
Convalescent plasma being a blood product may cause various transfusion reactions, such as allergic reactions and lung damage.
Some patients may face mild or no complications, while others may face more severe and even life-threatening complications.
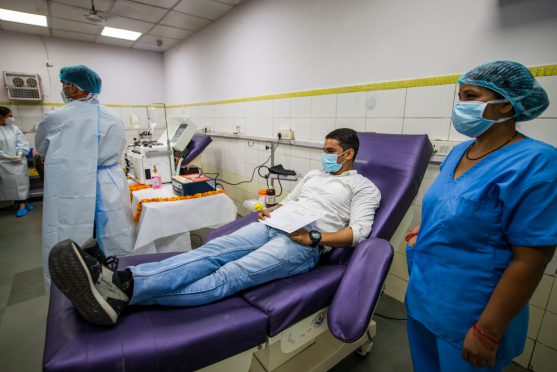
If You Have Recovered from COVID-19, how Safe is Plasma Donation?
The donors donate blood like any blood donation procedure. Plasma is separated from the cellular component of blood before being transfused in patients.
Earlier there have been calls from ICMR (Indian Council of Medical Research) and various state governments asking people who have recovered from COVID-19 to step out and volunteer and donate their plasma to help treat critically ill people.
The latest guidelines on management of COVID-19 in adults do not recommend use of convalescent plasma across any subgroup of patients.
NOTE: Please note that plasma therapy is still an investigational therapy. We cannot use it for all patients until it is proved to be safe and useful for regular use for all patients.


